Resource Hub
Tags: Diagnostics, In Vitro Diagnostics (IVD), IVD
Latest CDC Findings: A Wake‑Up Call for the $1.3 Billion Global Sepsis Diagnostics Market
Kalorama Information estimates that the global market for sepsis diagnostics reached approximately $1.3 billion in 2025, reflecting the enormous clinical and economic weight sepsis places on global healthcare. Yet despite steady growth and widespread use of diagnostic tools, sepsis is persistently one of the most difficult conditions to identify and manage. That reality is underscored by recent insights from the Centers for Disease Control and Prevention (CDC), which link sepsis to nearly one in five pediatric hospital deaths in the United States, highlighting both the limits of current sepsis diagnostic practices and the urgency of earlier detection.
According to the MSN.com article, CDC study links sepsis to nearly 1 in 5 pediatric hospital deaths, the CDC study analyzed data from more than 3.9 million pediatric hospitalizations throughout the U.S. over several years and found that sepsis occurred in a surprising number of cases and was involved in a notable share of in‑hospital pediatric deaths. Many of the affected children experienced severe complications, reinforcing how quickly sepsis can progress once it develops.
These pediatric findings mirror broader patterns identified by the CDC across the U.S. healthcare system. Annually, at least 1.7 million U.S. adults and more than 18,000 children develop sepsis. Roughly one in three adults who die in U.S. hospitals had sepsis during their hospital stay, while about one in five children who die in hospital were also affected. Among children who develop sepsis, approximately one in ten do not survive their hospitalization. In total, more than 350,000 adults and over 1,800 children with sepsis either die during hospitalization or are discharged to hospice care every year.
Sepsis Begins Before the Hospital
Arguably one of the CDC study’s most important observations is that most sepsis cases begin before a patient is even admitted to the hospital. Nearly a quarter to a third of individuals who develop sepsis had a healthcare visit in the week prior to hospitalization, and most had at least one underlying medical condition, such as chronic lung disease or a compromised immune system.
For pediatric care, this shifts some of the focus beyond inpatient treatment to earlier recognition in outpatient and emergency settings. It’s paramount that parents be aware of sepsis symptoms, as early signs frequently appear before hospital admission. For healthcare providers, these findings reinforce the need for vigilance when evaluating patients with evolving infections, especially those with recent illnesses or healthcare exposure.
Once sepsis is suspected, immediate evaluation and treatment are critical. Effective management typically involves rapid initiation of antibiotics, support of blood flow to vital organs, and, in some cases, surgical intervention to remove infected or damaged tissue.
Why Diagnosing Sepsis Remains Difficult: No Single Test Tells the Whole Story
Diagnosing sepsis remains complex even after decades of research and clinical focus. Sepsis is not a single disease but a dysregulated immune response to infection, which can present differently across patients and progress rapidly. Common clinical indicators of sepsis include fever, elevated heart rate, low blood pressure, and respiratory distress, but these signs overlap with many other acute conditions.
As Kalorama Information’s research shows, sepsis diagnosis typically relies on a combination of clinical assessment and multiple diagnostic tests, rather than a single definitive assay. Blood cultures remain a gold standard for pathogen identification, but their long turnaround times limit their usefulness during the early, most critical phases of care.
To complement cultures, laboratories rely heavily on in vitro diagnostics (IVD), particularly biomarkers such as procalcitonin (PCT), lactate, and C‑reactive protein (CRP), along with complete blood counts (CBCs). Molecular diagnostics, including PCR‑based assays and mass spectrometry technologies like MALDI‑TOF, are increasingly used to accelerate pathogen identification once cultures become positive. Medical imaging further supports clinicians by helping pinpoint sources of infection, though imaging itself is not specific to sepsis.
This multi‑layered approach speaks to the inherent difficulty of sepsis detection. No single test or technology can capture the full picture, forcing clinicians to synthesize incomplete data under intense time pressure.
A Fragmented Market Serving a Costly Condition
From a market perspective, sepsis remains unusual. There is no singular “sepsis test,” and most diagnostics used in sepsis management serve multiple clinical purposes. While specialized products are beginning to emerge, the majority of sepsis testing relies on broadly applicable tools used across infectious disease care.
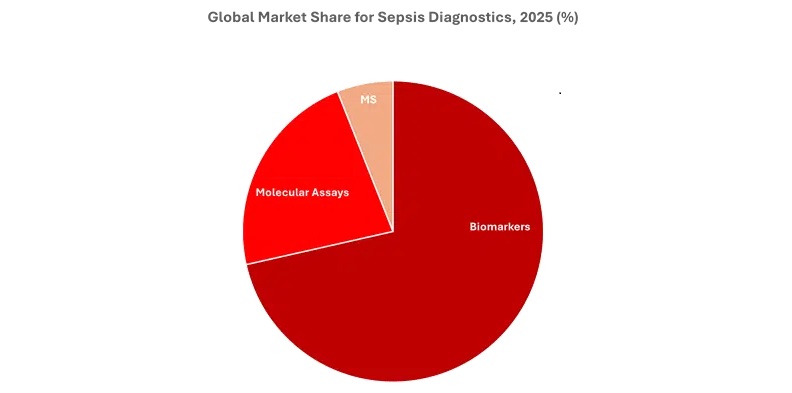
As previously mentioned, Kalorama Information estimates that biomarker assays, molecular diagnostics, and mass spectrometry systems used in sepsis diagnosis globally generated roughly $1.3 billion in sales in 2025. Biomarkers accounts for the majority of the overall sepsis diagnostics market, followed by molecular assays, and then mass spectrometry. Despite being the smallest segment, mass spectrometry for sepsis diagnostics is forecast to experience the largest compound annual growth rate (CAGR) of any segment from 2025–2031, growing at a double-digit rate.
The overall sepsis diagnostic market is projected to experience a mid single‑digit CAGR by 2031. Growth has been steady not rapid, reflecting both persistent clinical demand and slow adoption of newer technologies.
Herein lies the paradox. Sepsis is the most expensive condition treated in U.S. hospitals, yet diagnostic innovation often faces obstacles such as reimbursement uncertainty, workflow disruption, high upfront costs, and entrenched clinical practices. Even promising technologies frequently struggle to achieve widespread uptake.
What’s Next for Sepsis Diagnostics
Kalorama Information’s research emphasizes that progress in sepsis diagnosis almost certainly will not come from a single breakthrough technology. Instead, improvements will depend on how effectively laboratories and clinicians integrate multiple diagnostic approaches, supported by ongoing advances in biomarkers, molecular methods, and emerging data‑driven tools.
For a worldwide market already exceeding $1 billion annually, the issue is not demand, but rather it is coordination, adoption, and confidence. As pediatric outcomes in particular bring renewed attention to sepsis, the challenge for diagnostics remains the same: translating technological capability into earlier, more reliable decisions when time matters most.
Where to Learn More About Sepsis
Kalorama Information’s Worldwide Sepsis Diagnostic Products: Market Insights and Forecasts to 2031 delivers a comprehensive assessment of the worldwide market for sepsis diagnostic products, including in vitro diagnostics (IVD), molecular diagnostics, and related testing approaches. The report provides global sales data and projections through 2031 for major world regions and for key individual markets.

